Abstract
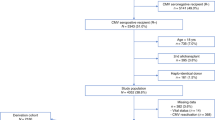
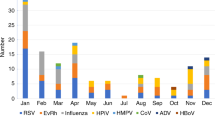
The objective of the study was the analysis of clinical types, outcomes, and risk factors associated with the outcome of adenovirus (ADV) infection, in children and adults after allo-HCT. A total number of 2529 patients (43.9% children; 56.1% adults) transplanted between 2000 and 2022 reported to the EBMT database with diagnosis of ADV infection were analyzed. ADV infection manifested mainly as viremia (62.6%) or gastrointestinal infection (17.9%). The risk of 1-year mortality was higher in adults (p = 0.0001), and in patients with ADV infection developing before day +100 (p < 0.0001). The 100-day overall survival after diagnosis of ADV infections was 79.2% in children and 71.9% in adults (p < 0.0001). Factors contributing to increased risk of death by day +100 in multivariate analysis, in children: CMV seropositivity of donor and/or recipient (p = 0.02), and Lansky/Karnofsky score <90 (p < 0.0001), while in adults: type of ADV infection (viremia or pneumonia vs gastrointestinal infection) (p = 0.0004), second or higher HCT (p = 0.0003), and shorter time from allo-HCT to ADV infection (p = 0.003). In conclusion, we have shown that in patients infected with ADV, short-term survival is better in children than adults. Factors directly related to ADV infection (time, clinical type) contribute to mortality in adults, while pre-transplant factors (CMV serostatus, Lansky/Karnofsky score) contribute to mortality in children.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Data represent the results of registry-based study of patients with documented ADV infection. No personal data are included. With the retrospective registry-based design of the study, no ethical review board approval was necessary. The data set remains the property of EBMT. It can be available upon reasonable request to the corresponding author.
References
Lindemans CA, Leen AM, Boelens JJ. How I treat adenovirus in hematopoietic stem cell transplant recipients. Blood. 2010;116:5476–85.
Matthes-Martin S, Feuchtinger T, Shaw PJ, Engelhard D, Hirsch HH, Cordonnier C, et al. European guidelines for diagnosis and treatment of adenovirus infection in leukemia and stem cell transplantation: summary of ECIL-4 (2011). Transpl Infect Dis 2012;14:555–63.
Lion T. Adenovirus infections in immunocompetent and immunocompromised patients. Clin Microbiol Rev. 2014;27:441–62.
Hiwarkar P, Kosulin K, Cesaro S, Mikulska M, Styczynski J, Wynn R, et al. Management of adenovirus infection in patients after haematopoietic stem cell transplantation: state-of-the-art and real-life current approach: a position statement on behalf of the Infectious Diseases Working Party of the European Society of Blood and Marrow Transplantation. Rev Med Virol. 2018;28:e1980.
Cesaro S, Berger M, Tridello G, Mikulska M, Ward KN, Ljungman P, et al. A survey on incidence and management of adenovirus infection after allogeneic HSCT. Bone Marrow Transplant. 2019;54:1275–80.
Czyzewski K, Styczynski J, Giebel S, Fraczkiewicz J, Salamonowicz M, Zajac-Spychala O, et al. Age-dependent determinants of infectious complications profile in children and adults after hematopoietic cell transplantation: lesson from the nationwide study. Ann Hematol. 2019;98:2197–211.
Styczynski J. Looking to the CAR-T future: vaccination, outpatient therapy, artificial intelligence and expanding indications. Acta Haematol Pol. 2022;53:151–52.
Kampouri E, Little JS, Rejeski K, Manuel O, Hammond SP, Hill JA. Infections after chimeric antigen receptor (CAR)-T-cell therapy for hematologic malignancies. Transpl Infect Dis. 2023:e14157 https://doi.org/10.1111/tid.14157.
Zajac-Spychala O, Pieczonka A, Wachowiak J, Fraczkiewicz J, Salamonowicz M, Kalwak K, et al. Adenovirus infection among pediatric patients with cancer and in pediatric recipients of hematopoietic stem cell: a multicenter nationwide study. J Med Virol. 2020;92:3187–93.
Papanicolaou GA, Dvorak CC, Dadwal S, Maron G, Prasad VK, Giller R, et al. Practice patterns and incidence of adenovirus infection in allogeneic hematopoietic cell transplant recipients: multicenter survey of transplant centers in the United States. Transpl Infect Dis. 2020;22:e13283.
Gonzalez-Vicent M, Verna M, Pochon C, Chandak A, Vainorius E, Brundage T, et al. Current practices in the management of adenovirus infection in allogeneic hematopoietic stem cell transplant recipients in Europe: the AdVance study. Eur J Haematol. 2019;102:210–17.
Sedlacek P, Petterson T, Robin M, Sivaprakasam P, Vainorius E, Brundage T, et al. Incidence of adenovirus infection in hematopoietic stem cell transplantation recipients: findings from the AdVance Study. Biol Blood Marrow Transplant. 2019;25:810–18.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation. 1974;18:295–304.
Iacobelli S. Suggestions on the use of statistical methodologies in studies of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 2013;48:S1–37.
Klein JP, Moeschberger ML. Survival analysis: techniques for censored and truncated data. New York: Springer-Verlag; 2003.
Schemper M, Smith TL. A note on quantifying follow-up in studies of failure time. Control Clin Trials. 1996;17:343–6.
Al-Heeti OM, Cathro HP, Ison MG. Adenovirus infection and transplantation. Transplantation. 2022;106:920–27.
Cesaro S. Adenovirus infection in allogeneic hematopoietic cell transplantation. Transpl Infect Dis. 2023;25:e14173.
Passweg JR, Baldomero H, Ciceri F, Corbacioglu S, de la Camara R, Dolstra H, et al. Hematopoietic cell transplantation and cellular therapies in Europe 2021. The second year of the SARS-CoV-2 pandemic. A Report from the EBMT Activity Survey. Bone Marrow Transplant. 2023;58:647–58.
Ljungman P, Brand R, Einsele H, Frassoni F, Niederwieser D, Cordonnier C. Donor CMV serologic status and outcome of CMV-seropositive recipients after unrelated donor stem cell transplantation: an EBMT megafile analysis. Blood. 2003;102:4255–60.
Ljungman P, Tridello G, Pinana JL, Ciceri F, Sengeloev H, Kulagin A, et al. Improved outcomes over time and higher mortality in CMV seropositive allogeneic stem cell transplantation patients with COVID-19; An infectious disease working party study from the European Society for Blood and Marrow Transplantation registry. Front Immunol. 2023;14:1125824.
Pikkel-Geva HZ, Grisariu S, Rivkin M, Stepensky P, Strahilevitz J, Averbuch D, et al. High rate of adenovirus detection in gastrointestinal biopsies of symptomatic stem cell transplant recipients. Clin Transplant. 2023;37:e15098.
Cesaro S, Porta F. Adenovirus infection in pediatric hematopoietic cell transplantation: a challenge still open for survival. J Clin Med. 2022;11:4827.
Styczynski J, Tridello G, Koster L, Knelange N, Wendel L, van Biezen A, et al. Decrease of lethal infectious complications in the context of causes of death (COD) after hematopoietic cell transplantation: COD-2 and COD-1 study of the Infectious Diseases Working Party EBMT. Bone Marrow Transplant. 2023;58:881–92.
Deambrosis D, Davies E, Turner A, Guiver M, Bonney D, Campbell H, et al. Burden of adenoviraemia predicts survival in paediatric recipients of allogeneic haematopoietic stem cell transplant. J Clin Virol. 2020;127:104373.
Lee YJ, Fang J, Zavras PD, Prockop SE, Boulad F, Tamari R, et al. Adenovirus viral kinetics and mortality in ex vivo T cell-depleted hematopoietic cell transplant recipients with adenovirus infection from a single center. J Infect Dis. 2020;222:1180–87.
Inamoto Y, Takeda W, Hirakawa T, Sakaguchi H, Nakano N, Uchida N, et al. Adenovirus disease after hematopoietic cell transplantation: a Japanese transplant registry analysis. Am J Hematol. 2022;97:1568–79.
Author information
Authors and Affiliations
Contributions
JS, MM – design of the study; NK, LW, GT – preparing database; GT – statistical analysis, tables, and figures; JS – writing manuscript; JS, MM, DA, SC, LG, PL, RdlC – analysis and critical revision of the paper; all authors – providing clinical data, revision, and final approval of the paper.
Corresponding author
Ethics declarations
Competing interests
MM reports lecture fees and board meeting fees from Allovir, bioMérieux, Gilead, Janssen, Moderna, Mundipharma, Pfizer, all outside the submitted work. JAS declares consultancy for Medac, Jazz, Vertex, and Kiadis Pharma in previous 24 months. All other authors declared no conflict of interest.
Ethics approval and consent to participate
The study was conducted in accordance with the EBMT Guidelines for Retrospective Studies and the principles of the Declaration of Helsinki. EBMT centres commit to obtain informed consent with the local regulations applicable at the time of transplantation in order to report pseudoanonymised data to the EBMT.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Styczynski, J., Tridello, G., Knelange, N. et al. Adenovirus infections after allogeneic hematopoietic cell transplantation in children and adults: a study from the Infectious Diseases Working Party of the European Society for Blood and Marrow Transplantation. Bone Marrow Transplant 59, 1402–1412 (2024). https://doi.org/10.1038/s41409-024-02361-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-024-02361-9